Purity: because the formula doesn't tell the whole story
In a nanoparticle, both phase purity and compositional purity must be assessed.
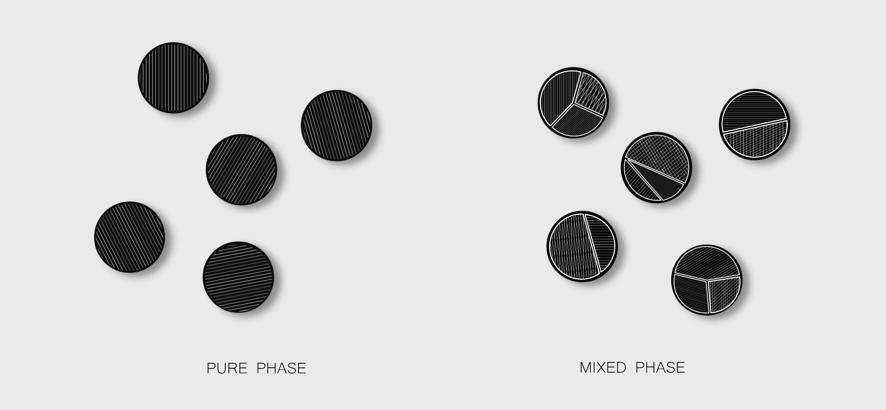
Phase purity refers to the number of distinct crystalline phases of the same composition that can be detected in a material. For instance, TiO₂ can be prepared in at least three different atomic arrangements with significantly different properties: rutile, anatase and brookite. Detection is not straightforward, as the chemical composition is identical across phases, and it requires specific characterization techniques such as X-ray Diffraction or Photoelectron Spectroscopy. The empirical formula, in other words, tells only part of the story.
Compositional purity refers to the presence of other, typically undesirable species such as contaminants. These may be inert in some applications while critical in others, even at trace quantities. Although they cannot be entirely eliminated, they must be minimised. Their origin can usually be traced to raw materials, processing cross-contamination, the environment, handling, or corrosion and abrasion of pipework and containers.
Understanding both the application and the nanomaterial is the most effective starting point for preventing any purity-related issue.












